Proposed Proton Pumping Mechanism and Catalytic Cycle of Cytochrome c Oxidase
Based on MD simulations, the free energy calculations, the combined DFT/continuum electrostatic solvation and pKa calculations,
we proposed the following mechanism of proton pumping in CcO.
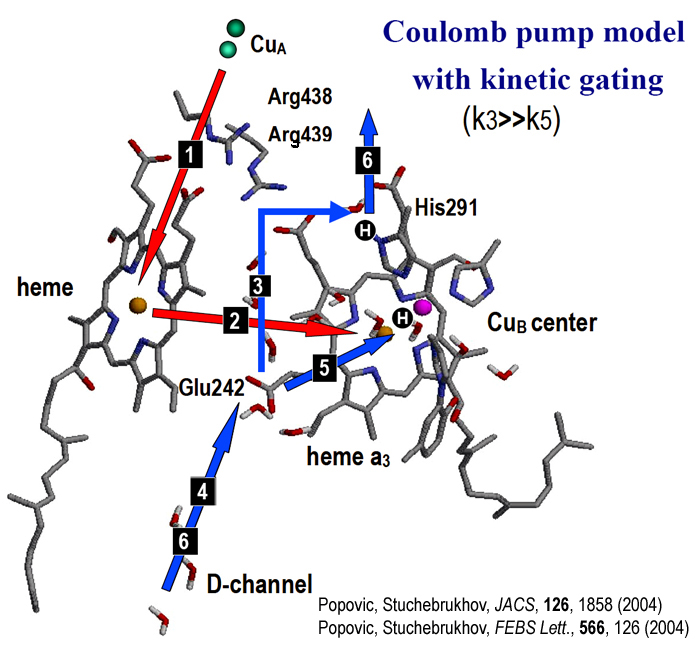
Upon ET from CuA to heme a (step 1), and subsequent ET from heme a to the BNC to form RR fully reduced state of BCN (step 2),
according to our model the pKa of His291 become around 17, and in step 3, a coupled PT from Glu to His occurs.
Step 4 is reprotonation of Glu (which pKa is always > 7), followed by the PT from Glu to BNC
(transfer of chemical proton, step 5). Due to a repulsive interaction between the two protons, a proton from
His-site is expelled in step 6, according to our calculations. Finally, last step in the sequence is repotonation
of Glu once again.
The main assumption of this model is that the rate k3 needs to be much higher than rate k5,
as indeed recent experimental observations may suggest.
The Proposed Catalytic Cycle of CcO
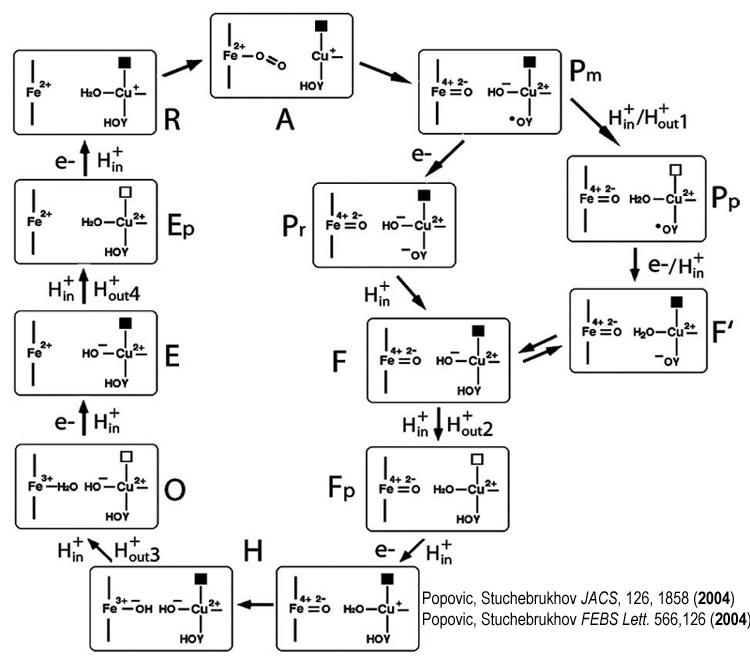
Recently, we proposed the catalytic cycle of CcO. The enzyme undergoes the complex redox and protonation changes (the state of the BNC is only shown) passing through different states of the catalytic cycle (R, A, P, F, H, O, E, R). Protons are pumped during S to Sp steps.
Scheme of redox-linked proton pumping involves the CuB His291 ligand
The pumping mechanism is based on ET reaction between two hemes that is coupled to a transfer of two protons. Upon ET, the first proton (fast reaction) is transferred to the PLS (His291), while subsequent transfer of the second "chemical" proton to the binuclear center (slow reaction) is accompanied by the ejection of the first (pumped) proton.
Kinetic gating, absence of a mechanical gate
We argue that it is here the energetics of proton translocation along the channels, and redox-dependent affinity switches, that explain uni-directionality of proton translocation along the channels, rather than conformational changes usually assumed in the pumping models.